 * Some caveats: for example, aluminium will react slowly with water, if the thin aluminium oxide layer that prevents it from reacting is damaged. * This graphic doesn’t contain every metal in the periodic table. Majority of metals occur naturally in compounds, which we must remove them from. Some metals are so unreactive they occur largely uncombined with other elements, simple to obtain. And so once again, the alkaline earth metals are going to react in similar ways. 
You find them in combination with other elements. At the top of the list are the highly reactive metals that lose electrons during a chemical reaction to form ions. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. 
This is because they can react with the compounds in metal ores, and displace the metals, aiding with their extraction. Your alkaline earth metals are reactive- not quite as reactive as the metals in group 1, but you dont find these in the pure state, either. The reactivity series of metals is a list of metals arranged in their order of reactivity from highest to lowest. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. Carbon and hydrogen are also shoehorned in between entries in the list, despite being non-metals. Copper sulfate + zinc → zinc sulfate + copper Magnesium sulfate + zinc → NO REACTION The reactivity series also gives us an insight into why different metals are extracted from their ores in different ways. Conversely, if we react a metal with another metal lower in the series, no reaction will take place. The more reactive metal will take the place of the less reactive metal in the compound. Metals are shiny, and they conduct heat and electricity. Notice how metals are on the left and bottom of the periodic table, while non-metals are on the right and top. If a metal compound reacts with a metal that’s above it in the reactivity series, a displacement reaction will occur. soft, less reactive metals, valence 2, almost always M 2+ ions: 13: Boron Group: non-metals and metals, valence 3: 14: Carbon Group. Uses of this: * predict the outcome of certain chemical reactions. In this video (Part 1 of 2), we take a look at metals, wh. Transition metals are much less reactive Gold and platinum have little in the way of chemical reaction. In this short series, we look at what makes certain elements really reactive and others just.not. Closely followed by the marginally less reactive group 2 metals. The largest atomic radius of elements in their period. Metals have a range of reactivities: Videos: the classic alkali metals in water demonstration The reactivity series offers a ranking of the metals in order of their reactivity. Highly reactive, with reactivity increasing moving down the group. are oxidized) when they undergo chemical reactions They normally do not accept electrons. They readily form alloys and lose electrons to form stable cations.The text below has been excerpted from Compound Chemistry Student handout: Reactivity Series of Metals (PDF) This graphic places a selection of common metals into order of reactivity, as well as showing their reactions with air, water and steam. Other chemical properties include: Electropositive Character: Metals tend to have low ionization energies, and typically lose electrons (i.e. 
They are almost all hard, high-melting solids that conduct heat and electricity well. The elements in group 3 have an ns 2 (n 1)d 1 configuration, except for lawrencium (Lr): its 7s 2 7p 1 configuration exceptionally does not fill the 6d orbitals at all. The transition elements have many properties in common with other metals. In the periodic table, the transition metals are present in ten groups (3 to 12). There is no unique and fully consistent way to define the reactivity series, but it is common to use the three types of reaction listed below, many of which can be performed in a high-school laboratory (at least as demonstrations). Increasing the supply of lanthanoid elements is one of the most significant challenges facing the industries that rely on the optical and magnetic properties of these materials. In 2008, dysprosium cost $110/kg by 2014, the price had increased to $470/kg. A computer hard drive is shown with a metallic component highlighted in a red circle.Īs the demand for lanthanide materials has increased faster than supply, prices have also increased. Closeup of a flat screen shows the many individual pixels which are red green and blue in color. (credit b: modification of work by “KUERT Datenrettung”/Flickr) A. (b) Neodymium magnets are commonly found in computer hard drives. 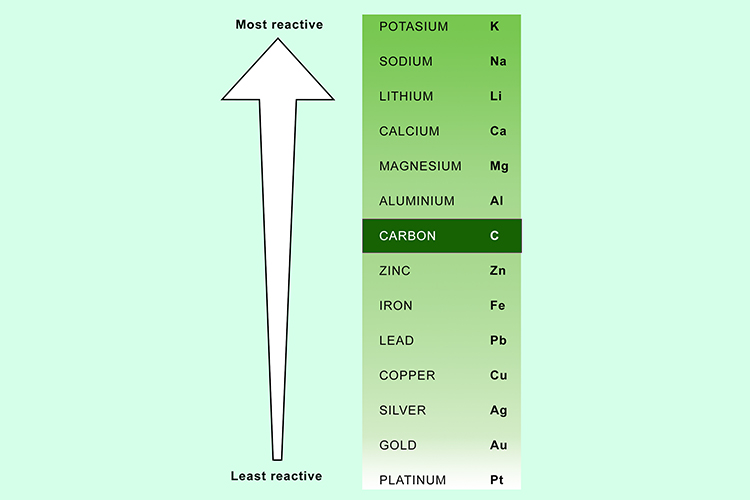
\): (a) Europium is used in display screens for televisions, computer monitors, and cell phones. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
